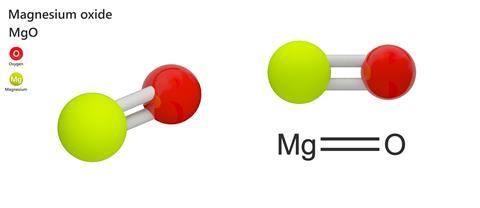A comparison of carbon footprints of magnesium oxide and magnesium hydroxide produced from conventional processes - ScienceDirect
![Amazon.com: Clean Formula Magnesium Oxide 400mg [High Potency] Supplement | No Harmful Additives - 120 Tablets : Health & Household Amazon.com: Clean Formula Magnesium Oxide 400mg [High Potency] Supplement | No Harmful Additives - 120 Tablets : Health & Household](https://m.media-amazon.com/images/S/aplus-media-library-service-media/da43a736-3bf6-4013-a182-d767d61766b4.__CR0,0,970,600_PT0_SX970_V1___.jpg)
Amazon.com: Clean Formula Magnesium Oxide 400mg [High Potency] Supplement | No Harmful Additives - 120 Tablets : Health & Household

Magnesium Oxide‐Assisted Dual‐Cross‐Linking Bio‐Multifunctional Hydrogels for Wound Repair during Full‐Thickness Skin Injuries - Tang - 2021 - Advanced Functional Materials - Wiley Online Library
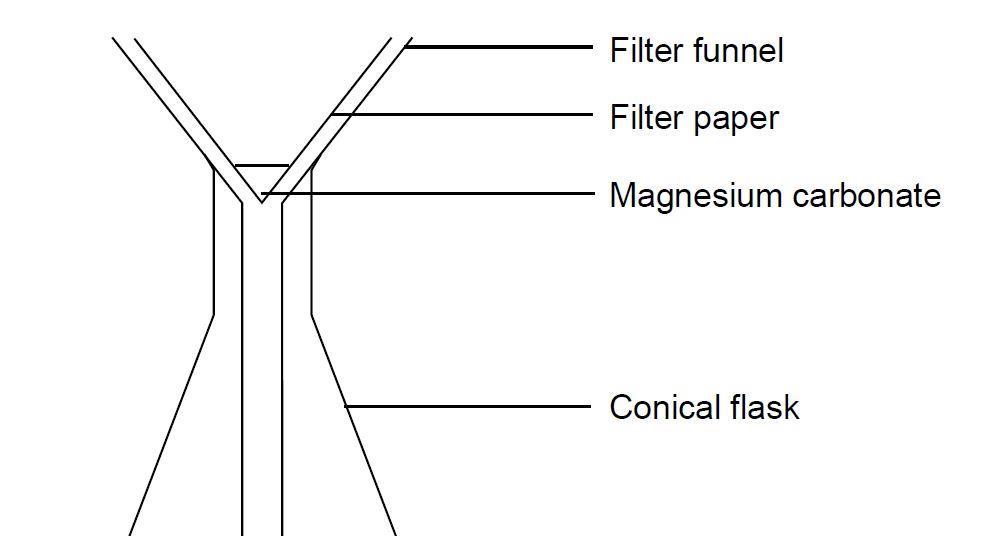
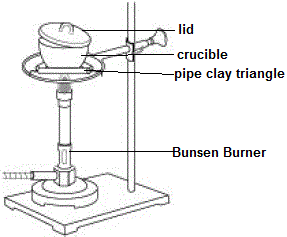
Magnesium and carbon dioxide – Student sheet Burning magnesium in carbon dioxide – what will happen? To do
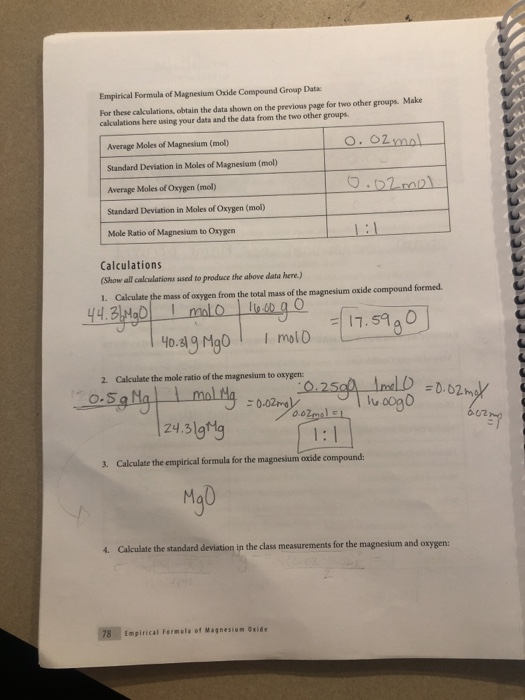
A 3.250g sample of magnesium is burned in a container of 12.500g oxygen. What mass of oxygen gas remains unreacted after the magnesium has been completely consumed to form magnesium oxide as